Mercury is a potent neurotoxin present in our daily lives and our body can accumulate it over the years. Food consumption, such as fish and rice, is the most common source of mercury exposure. Mercury can be found in dental amalgams, compact fluorescence lamps, vaccines, drugs, and electronics or can be used in artisanal gold mining. Finding the source of mercury contamination in our bodies is crucial for treatment and forensic investigations, but at present knowledge of the molecular form of mercury in human tissues and fluids, which could indicate source, is limited.
However, an international team from the CNRS, the Universities of Grenoble Alpes, Bordeaux and Franche‐Comté, France, the ESRF, the European synchrotron in Grenoble, France and the University of Illinois at Chicago, US, has developed new analytical capabilities that allow the identification of chemical forms of mercury in human hair that trace exposure. These results are published in Environmental Science & Technology.
Background:
Mercury contamination, a public health issueMercury is considered by the World Health Organisation (WHO) as one of the top ten chemicals or groups of chemicals of major public health concern. Exposure to mercury even small amounts may cause serious health problems in the nervous, digestive and immune systems, and is particularly dangerous for very young children and development of the child in utero. Identifying the type of mercury contamination, the dose, and the period and duration of exposure could provide key indicators to treat mercury poisoning.
The new study:
New analytical capabilities to identify chemical forms of mercury in human hairUntil today, depending on the suspected source of contamination, mercury intake has been monitored by measurement of mercury concentration in urine, blood, or scalp hair. These measurements help to diagnose the dose of poisoning and provide data for epidemiological studies, but provide incomplete information on the source of mercury exposure essential for treatment and forensic investigations. Although urinary mercury concentration is considered to be the most accurate and widely used biomarker for assessing chronic exposure to mercury vapour and divalent mercury, we showed that inorganic mercury from dental amalgams can be detected in hair with distinct intermolecular structure from that of methylmercury from fish consumptionh says Jean-Paul Bourdineaud, Professor of environmental toxicology at the University of Bordeaux, France.
Epidemiological studies on mercury intake through fish consumption assume that hair concentration is an indicator of only this source. Our results show that this assumption may not always be true says Kathryn Nagy, Professor of Earth and Environmental Sciences at the University of Illinois at Chicago. With an average growth rate of about 1 cm per month, hair can capture contamination events with high time resolution. The team of researchers developed new analytical capabilities that allowed them to identify chemical forms of mercury in human hair. Using new synchrotron instrumentation, they found that mercury can be linked to source of exposure by precise characterisation of its bonding environment. Experiments at the ESRF have shown, for example, that a mercury spike along a hair strand was correlated, in this particular case, with a specific unsafe removal of dental amalgam. The data provide signatures of mercury sources, distinguish exogenous vs. endogenous exposure to inorganic mercury, and indicate the timing to within 1 or 2 days of an exposure event.
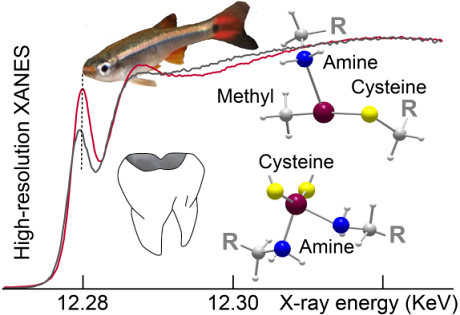
Fig.1: Spectral signature of two chemical forms of
mercury in hair, one originating from fish consumption and the other
from removal of a dental amalgam. In the first case (fish), mercury is
methylated and bonded to one cysteine sulfur and one amine group from
hair proteins (mostly keratin). In the second (dental amalgam), mercury
is bonded to two cysteine sulfurs and two amine groups from hair
proteins. Credit: @A.Manceau
The technical challengesAs straightforward as it may seem, the team had to deal with some technical challenges before managing to retrieve this information: structural characterisation is complicated by the low concentration of mercury typical of most of the general populationfs hair (typically 0.1 to 3 ng of mercury/mg hair), and the flexibility of the mercury bonding environment which exhibits diverse coordination arrangements with carbon, nitrogen, oxygen, and sulfur atoms. "When we realized that the microscopic 2.5 ng mercury hot-spot had a specific molecular signature, we were amazed. Still, to preclude any ambiguity on its origin, we had to determine the form of mercury and how it was chemically bonded to the hair proteins. This took several months of intense data analysis and calculations" says Alain Manceau, Director of Research at the CNRS. To address these challenges, the team used the high performance capabilities of the ESRF by exploiting the X-ray nanoprobe experimental station at ID16B, an ESRF nano-analysis beamline, and by constructing a high-luminosity X-ray crystal multi-analyser that fully takes advantage of the high-brilliance of the ESRF ID26 spectrometer beamline. As explained by Remi Tucoulou, scientist at ID16B, and Pieter Glatzel, scientist at ID26: Such measurements on samples with very low metal concentrations require instruments that are extremely sensitive and at the same time provide very high resolution for chemical characterisation.
Story Source: Materials provided by
CNRS (Délégation Paris Michel-Ange). Note: Content may be edited for style and length.
 The original study
The original study:

A. Manceau, M. Enescu, A. Simionovici, M. Lanson, M. Gonzalez-Rey, M. Rovezzi, R. Tucoulou, P. Glatzel, K.L. Nagy, J.P. Bourdineaud,
Chemical forms of mercury in human hair reveal sources of exposure. Environ. Sci. Technol.
doi: 10.1021/acs.est.6b03468
 Related studies (newest first):
Related studies (newest first):

A. Manceau, C. Lemouchi, M. Rovezzi, M. Lanson, P. Glatzel, K.L. Nagy, I. Gautier-Luneau, Y. Joly, M. Enescu.
Structure, bonding, and stability of mercury complexes with thiolate and thioether ligands from high-resolution XANES spectroscopy and first-principles calculations. Inorg. Chem., 54 (2015) 11776-11791.
doi: 10.1021/acs.inorgchem.5b01932

A.
Manceau, C. Lemouchi, M. Enescu, A,-C. Gaillot, M. Lanson, V. Magnin,
P. Glatzel, B.A. Poulin, J.N. Ryan, George R. Aiken, I.
Gautier-Luneau,K.L. Nagy.
Formation of mercury
sulfide from Hg(II)-thiolate complexes in natural organic matter. Environ. Sci. Technol. 2015, 49, 9787-9796.
doi: 10.1021/acs.est.5b02522
M.L. Li, L.S. Sherman, J.D. Blum, P. Grandjean, B. Mikkelsen, P. Weihe, E.M. Sunderland, J.P. Shine,
Assessing sources of human methylmercury exposure using stable mercury isotopes. Environ. Sci. Technol., 48 (2014) 8800-8806.
doi: 10.1021/es500340r

L.S. Sherman, J.D. Blum, A. Franzblau, N. Basu,
New insight into biomarkers of human mercury exposure using naturally occurring mercury stable isotopes. Environ. Sci. Technol., 47 (2013) 3403-3409.
doi: 10.1021/es305250z

L. Laffont, J.E. Sonke, L. Maurice, S.L. Monrroy, J. Chincheros, D. Amouroux, P. Behra,
Hg speciation and stable isotope signatures in human hair as a tracer for dietary and occupational exposure to mercury. Environ. Sci. Technol., 45 (2011) 9910-9916.
doi: 10.1021/es202353m

J.G. Dórea, V. Bezerra, V. Fajon, M. Horvat,
Speciation of methyl- and ethyl-mercury in hair of breastfed infants acutely exposed to thimerosal-containing vaccines. Clin. Chim. Acta, 412 (2011) 1563-1566.
doi: 10.1016/j.cca.2011.05.003

Graham N. George, Aatya P. Singh, Gary J. Myers, Gene E. Watson, Ingrid J. Pickering,
The chemical forms of mercury in human hair: a study using X-ray absorption spectroscopy, J. Biol. Inorg. Chem., 15/5 (2010) 709-715.
doi: 10.1007/s00775-010-0638-x

H. Fakour, A. Esmaili-Sari, F. Zayeri,
Scalp hair and saliva as biomarkers in determination of mercury levels in Iranian women: Amalgam as a determinant of exposure. J. Hazard. Mater., 177 (2010) 109-113.
doi: 10.1016/j.jhazmat.2009.12.002
M.A. McDowell, C.F. Dillon, J. Osterloh, P.M. Bolger, E. Pellizzari, R. Fernando, R.M. de Oca, S.E. Schober, T. Sinks, R.L. Jones, K.R. Mahaffey,
Hair mercury levels in US children and women of childbearing age: Reference range data from NHANES 1999-2000. Environ. Health Persp., 112 (2004) 165-1171.
doi: 10.1289/ehp.7046
H.H. Harris, I.J. Pickering, G.N. George,
The chemical form of mercury in fish. Science, 301 (2004) 1203.
doi: 10.1126/science.1085941
S.W. Lindow, R. Knight, J. Batty, S.J. Haswell,
Maternal and neonatal hair mercury concentrations: the effect of dental amalgam. Br. J. Obstet. Gynaecol., 110 (2003) 287-291.
doi: 10.1046/j.1471-528.2003.02257.x 
D. Airey,
Total mercury concentrations in human-hair from 13 countries in relation to fish consumption and location. Sci. Total Environ., 31 (1983) 157-180.
doi: 10.1016/0048-9697(83)90067-0
 Related EVISA Resources
Related EVISA Resources
 Brief summary: Chemical speciation analysis for nutrition and food science
Brief summary: Chemical speciation analysis for nutrition and food science Brief summary: Speciation and Toxicity
Brief summary: Speciation and Toxicity  Link database: Toxicity of Organic mercury compounds
Link database: Toxicity of Organic mercury compounds  Link database: Human exposure to methylmercury via the diet
Link database: Human exposure to methylmercury via the diet
 Related EVISA News
Related EVISA News
 December 29, 2013: A new study finds: Inorganic mercury stays in the brain for years if not decades
December 29, 2013: A new study finds: Inorganic mercury stays in the brain for years if not decades  December
13, 2013: Most Canadians having dental amalgam in their mouth are
exposed to mercury at levels surpassing the reference exposure level
(REL)
December
13, 2013: Most Canadians having dental amalgam in their mouth are
exposed to mercury at levels surpassing the reference exposure level
(REL)  November 20, 2013: EPA Study: Mercury Levels in Women of Childbearing Age Drop 34 Percent
November 20, 2013: EPA Study: Mercury Levels in Women of Childbearing Age Drop 34 Percent  October 12, 2013: Minamata Convention is adopted
October 12, 2013: Minamata Convention is adopted  March 22, 2013: Mercury isotope fractionation provides new tool to trace the source of human exposure
March 22, 2013: Mercury isotope fractionation provides new tool to trace the source of human exposure  January 21, 2013: UNEP mercury treaty exempts vaccines for children
January 21, 2013: UNEP mercury treaty exempts vaccines for children
 January 14, 2013: Mercury Levels in Humans and Fish Around the World Regularly Exceed Health Advisory Levels
January 14, 2013: Mercury Levels in Humans and Fish Around the World Regularly Exceed Health Advisory Levels
 December 24, 2012: Mercury in food EFSA updates advice on risks for public health
December 24, 2012: Mercury in food EFSA updates advice on risks for public health December 9, 2012: Mercury in fish more dangerous than previously
believed; Scientists urge for effective treaty ahead of UN talks
December 9, 2012: Mercury in fish more dangerous than previously
believed; Scientists urge for effective treaty ahead of UN talks
 June 17, 2012: Factors Affecting Methylmercury Accumulation in the Food Chain
June 17, 2012: Factors Affecting Methylmercury Accumulation in the Food Chain
 August 21, 2009: USGS Study Reveals Mercury Contamination in Fish Nationwide
August 21, 2009: USGS Study Reveals Mercury Contamination in Fish Nationwide
 February 11, 2009: Mercury in Fish is a Global Health Concern
February 11, 2009: Mercury in Fish is a Global Health Concern
 October 30, 2008: Precautionary approach to methylmercury needed
October 30, 2008: Precautionary approach to methylmercury needed
 March 11, 2007: Methylmercury contamination of fish warrants worldwide public warning
March 11, 2007: Methylmercury contamination of fish warrants worldwide public warning
 February 9, 2006: Study show high levels of mercury in women related to fish consumption
February 9, 2006: Study show high levels of mercury in women related to fish consumption
 January 12, 2005: Number of fish meals is a good predictor for
the mercury found in hair of environmental journalists
January 12, 2005: Number of fish meals is a good predictor for
the mercury found in hair of environmental journalists
 April 27, 2004: FDA/EPA recommends pregnant women to restrict
their fish consumption because of methylmercury content
April 27, 2004: FDA/EPA recommends pregnant women to restrict
their fish consumption because of methylmercury content
last time modified: October 9, 2016