By using XAS imaging, researchers reveal the distribution of arsenic species in a hyperaccumulating fern in-vivo.
Background:Plants such as the fern Pteris vittata can take up toxic substances from the soil and store them in their fronds (leaves). Such hyperaccumulation makes these plants potentially attractive for cleaning up polluted sites by phytoremediation. Unfortunately, the understanding of uptake, transformation, transport and storage of the species involved is still limited, which hampers the optimization of the process e.g. by genetically modyfing plants.
The new study:The multidisciplinary research team around Ingrid J. Pickering mapped live plants and single-cell-thick gametophytes by hitting them with microscopic X-ray beams at different energy levels. By using X-ray absorption spectroscopy (XAS) and XAS imaging, information about the distribution of arsenic species in vivo could be obtained. Different arsenic forms show up at different energies, providing a whole picture, says Graham George of the University of Saskatchewan (Canada). When you do it this way, features kind of pop out of the map, says Ingrid Pickering, Georges colleague and the first author of the work.
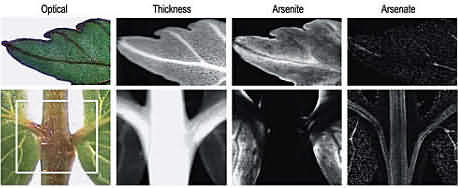 Reprinted with permission from Environ. Sci. Technol., 40 (2006) 5010-5014. Copyright (2006) American Chemical Society
Reprinted with permission from Environ. Sci. Technol., 40 (2006) 5010-5014. Copyright (2006) American Chemical SocietyThe images show that the fern transforms arsenate (AsO
43) into the even more toxic arsenite (H
2AsO
3) in its leaves, not in its roots as previously thought. Arsenic-thiolate species surrounding veins may be intermediates in this reduction. It also tucks the arsenite away inside its cell vacuoles, where it is stored at high concentrations; this may be a coping mechanism against arsenites toxicity.
Exactly how the plant transforms the arsenic remains unclear, but the researchers who captured the new pictures say that their technique (which uses synchrotron radiation at the Stanford Linear Accelerator Center) may be the most precise yet in showing where and in what forms the fern stores arsenic.
 The original study
The original study
Ingrid J. Pickering, Luke Gumaelius, Hugh H. Harris, Roger C. Prince, Gregory Hirsch, Jo Ann Banks, David E. Salt, Graham N. George,
Localizing the Biochemical Transformations of Arsenate in a Hyperaccumulating Fern, Environ. Sci. Technol., 40/16 (2006) 5010-5014.
DOI: 10.1021/es052559a Related studies
Related studies Kevin A. Francesconi
Kevin A. Francesconi, Pornsawan Visoottiviseth, Weeraphan Sridokchan,
Walter Goessler,
Arsenic species in an arsenic hyperaccumulating fern, Pityrogramma calomelanos: a potential phytoremediator of arsenic-contaminated soils, Sci. Total Environ., 284/1-3 (2002) 27-35.
DOI:10.1016/S0048-9697(01)00854-3 
Wei-hua Zhang, Yong Cai, Cong Tu, Lena Q. Ma,
Arsenic speciation and distribution in an arsenic hyperaccumulating plant, Sci. Total Environ., 300/1-3 (2002) 167-177.
DOI: 10.1016/S0048-9697(02)00165-1
Jun-ru Wang, Fang-Jie Zhao, Andrew A. Meharg, Andrea Raab,
Jörg Feldmann, Steve P. McGrath,
Mechanisms of Arsenic Hyperaccumulation in Pteris vittata. Uptake Kinetics, Interactions with Phosphate, and Arsenic Speciation, Plant Physiol., 130/3 (2002) 1552-1561.
DOI: 10.1104/pp.008185
D.E. Salt, R.C. Prince, I.J. Pickering,
Chemical speciation of accumulated metals in plants: evidence from X-ray absorption spectroscopy, Microchem. J., 71/2-3 (2002) 255-259.
DOI:10.1016/S0026-265X(02)00017-6
E. Lombi, F.J. Zhao, M. Fuhrmann, L.Q. Ma, S.P. McGrath,
Arsenic distribution and speciation in the fronds of the hyperaccumulator Pteris vittata, New Phytol., 156 (2002) 195-203.
DOI:10.1046/j.1469-8137.2002.00512.x
C. Tu, L.Q. Ma, W. Zhang, Y. Cai, W.G. Harris,
Arsenic species and leachability in the fronds of the hyperaccumulator Chinese brake (Pteris vittata L.), Environ. Pollut., 124/2 (2003) 223-230.
DOI: 10.1016/S0269-7491(02)00470-0
Wei-hua Zhang, Yong Cai,
Purification and characterization of thiols in an arsenic hyperaccumulator under arsenic exposure, Anal. Chem., 75/24 (2003) 7030-7035.
DOI: 10.1021/ac034697t
Y. Cai, J. Su, L.Q. Ma,
Low molecular weight thiols in arsenic hyperaccumulator Pteris vittata upon exposure to arsenic and other trace elements, Environ. Pollut., 129/1 (2004) 69-78.
DOI:10.1016/j.envpol.2003.09.020 
L. Gumaelius, B. Lahner, D.E. Salt, J.A. Banks,
Arsenic Hyperaccumulation in Gametophytes of Pteris vittata. A New Model System for Analysis of Arsenic Hyperaccumulation, Plant Physiol., 136/2 (2004) 3198-3208.
DOI: 10.1104/pp.104.044073
W. Zhang, Y. Cai, K.R. Downum, L.Q. Ma,
Arsenic complexes in the arsenic hyperaccumulator Pteris vittata (Chinese brake fern), J. Chromatogr. A, 1043/2 (2004) 249-254.
DOI: 10.1016/j.chroma.2004.05.090
W. Zhang, Y. Cai, K.R. Downum, L.Q. Ma,
Thiol synthesis and arsenic hyperaccumulation in Pteris vittata, (Chinese brake fern), Environ. Pollut., 131/3 (2004) 337-345.
DOI: 10.1016/j.envpol.2004.03.010
Zhe-Chun Huang, Tong-bin Chen, Mei Lei, Tian-Dou Hu,
Direct determination of arsenic species in arsenic hyperaccumulator Pteris vittatat by EXAFS, Acta Botanica Sinica, 46/1 (2004) 46-50.

Maria I. Silva Gonzaga, Jorge A.G. Santos, Lena Q. Ma,
Arsenic chemistry in the rhizosphere of Pteris vittata L. and Nephrolepis exaltata L., Environ. Pollut., 143/2 (2006) 254-260.
DOI: 10.1016/j.envpol.2005.11.037 
Akiko Hokura, Ryoko Omuma, Yasuko Terada, Nobuyuki Kitajima, Tomoko Abe, Hiroyuki Saito, Shigeo Yoshida, Izumi Nakai,
Arsenic distribution and speciation in an arsenic hyperaccumulator fern by X-ray spectrometry utilizing a synchrotron radiation source, J. Anal. At. Spectrom., 21/3 (2006) 321.
DOI: 10.1039/b512792k
Nandita Singh, Lena Q. Ma,
Arsenic speciation, and arsenic and phosphate distribution in arsenic hyperaccumulator Pteris vittata L. and non-hyperaccumulator Pteris ensiformis L.,
Environ. Pollut., 141/2 (2006) 238-246.
DOI: 10.1016/j.envpol.2005.08.050 Related EVISA News
Related EVISA News May 7, 2007: Arsenic-absorbing fern may soak up toxic metal to repel hungry bugs, UF researchers say
May 7, 2007: Arsenic-absorbing fern may soak up toxic metal to repel hungry bugs, UF researchers saylast time modified: June 21, 2020