Researchers at Yale University and University of Illinois at Chicago detail the molecular mechanisms that govern the metabolism of selenium in the human body.
Background:Selenium and its compounds are essential for animals and humans, but its physiological nature is ambivalent. It can cause disease by deficiency, but it is toxic at levels less than an order of magnitude above those required for optimum healthy conditions. Selenocysteine is the only genetically encoded amino acid in humans whose biosynthesis occurs on
its cognate transfer RNA (tRNA). O-Phosphoseryl-tRNA:selenocysteinyl-tRNA synthase (SepSecS) catalyzes the final step of selenocysteine formation by a poorly understood tRNA-dependent mechanism.
The new study:
In the July 17 issue of the journal Science, researchers at
Yale
University and
University of Illinois at Chicago detail the molecular
mechanisms that govern its metabolism in the human body.
"It must require an intricately regulated uptake system," said
Dieter Söll, co-senior author of the paper, Sterling Professor of Molecular Biophysics and Biochemistry at Yale. "There are 25 human selenoproteins, and most of them are probably essential for life."
Selenium is thought to offer protection from diverse human ailments including adverse mood states, cardiovascular disease, viral infections and cancer.
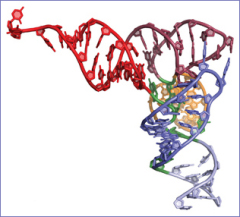
Selenocysteine is the most active metabolite of selenium in humans. It is unique among amino acids because it is the only one synthesized directly on a transfer RNA (tRNA) molecule, which shuttles the amino acids to the protein-making machinery within cells. Proteins that contain selenocysteine are responsible for recycling protective anti- oxidants such as vitamin C and coenzyme Q10.
Figure: Selonocysteine tRNA moleculeSöll's team for the first time captured images of how selenocysteine is created on a super-sized tRNA molecule, which seems to have a highly specialized role in nature. The 20 other amino acids and their associated tRNAs use the same protein vehicle, called an elongation factor, for transport to the ribosome. However, nature has provided this large tRNA molecule with a specialized elongation factor that chauffeurs only selenocysteine to the ribosome.
"This structure reveals most aspects of the mechanism for the formation of selenocysteine and provides an answer to 20 years of biochemical work in the field," said Sotiria Palioura, lead author of the study and an M.D./Ph.D. candidate at Yale.
The findings may lead to greater understanding of autoimmune liver disease. The tRNA complex described in the Science paper is the target of antibodies in patients with Type 1 autoimmune hepatitis. "The region that the antibody is supposed to recognize is at the business end of this molecule, where we see the reaction happening," Palioura said.
"Selenocysteine has been found to be a critical component of enzymes involved in a number of normal and disease processes," said Michael Bender of the National Institutes of Health's National Institute of General Medical Sciences. "This basic study, which has shed light on selenocysteine's unique biosynthetic pathway, could ultimately have an impact on many aspects of human health, including the immune response, neurodegeneration, cardiovascular disease, and cancer."
Other Yale authors on the paper were R. Lynn Sherrer and Thomas A. Steitz. Senior co-author on the paper was Miljan Simonovic of the University of Illinois at Chicago.
Funding for the research was provided by the National Institute for General Medical Sciences, the Department of Energy, and the Howard Hughes Medical Institute at Yale University.
Source:
Yale University The cited study:
The cited study:
Sotiria Palioura, R. Lynn Sherrer, Thomas A. Steitz, Dieter Söll, Miljan Simonovic,
The Human SepSecS-tRNASec Complex Reveals the Mechanism of Selenocysteine Formation, Science, 325/5938 (2009) 321-325.
DOI: 10.1126/science.1173755 Related studies
Related studies
Caterina Di Cosmo, Neil McLellan, Xiao-Hui Liao, Kum Kum Khanna, Roy E.
Weiss, Laura Papp, Samuel Refetoff,
Clinical and molecular characterization of a novel selenocysteine insertion sequence-binding protein 2 (SBP2) gene mutation (R128X), Clin. Endocrinol. Metab. , published online July 14, 2009.
DOI: 10.1210/jc.2009-0686

M. Birringer, S. Pilawa, L. Flohe,
Trends in selenium biochemistry, Nat. Prod. Rep., 19 (2002) 693-718.
DOI: 10.1039/b205802m 
Dietrich Behne, A. Kyriakopoulos,
Mammalian selenium-containing proteins, Annu. Rev. Nutr., 21 (2001) 453-473.
DOI: 10.1146/annurev.nutr.21.1.453 
G.N. Schrauzer,
Selenomethionine: A Review of Its Nutritional Significance, Metabolism and Toxicity, J. Nutr., 130 (2000) 1653-1656.
DOI:
10.1093/jn/130.7.1653

T.C. Stadtman,
Selenosysteine, Annu. Rev. Biochem., 65 (1996) 83-100.
DOI: 10.1146/annurev.bi.65.070196.000503
A. Böck, K. Forchhammer, J. Heider, C. Baron,
Selenoprotein synthesis: an expansion of the genetic code, Trends Biochem. Sci., 16 (1991) 463-467.
DOI:
10.1016/0968-0004(91)90180-4
 Related EVISA Resources
Related EVISA Resources Link database: Protein-bound selenium and human health
Link database: Protein-bound selenium and human health Link Database: Toxicity of selenium
Link Database: Toxicity of selenium Brief summary: Chemival speciation analysis for the life sciences
Brief summary: Chemival speciation analysis for the life sciences
 Related EVISA News (newest first)
Related EVISA News (newest first)
 August 16, 2016: Toxicity and bioavailability of different selenium metabolites
August 16, 2016: Toxicity and bioavailability of different selenium metabolites
 July 21, 2015: Polish selenium supplements not always labeled accurately
July 21, 2015: Polish selenium supplements not always labeled accurately
 December 17, 2014: Decreased risk of colorectal cancer linked with higher selenium status
December 17, 2014: Decreased risk of colorectal cancer linked with higher selenium status
 August 9, 2011: New selenium metabolites found in human serum
August 9, 2011: New selenium metabolites found in human serum May 22, 2011: Does Selenium Prevent Cancer? It May Depend on Which Form People Take
May 22, 2011: Does Selenium Prevent Cancer? It May Depend on Which Form People Take
 May 12, 2011: Review: Selenium doesn't prevent cancer
May 12, 2011: Review: Selenium doesn't prevent cancer June 19, 2010: A
new Selenium-containing compound, Selenoneine, found as the predominant
Se-species in the blood of Bluefin Tuna
June 19, 2010: A
new Selenium-containing compound, Selenoneine, found as the predominant
Se-species in the blood of Bluefin Tuna  July 20, 2009: Researchers Reveal Selenium's Metabolism In Life-Giving Amino Acids
July 20, 2009: Researchers Reveal Selenium's Metabolism In Life-Giving Amino Acids October 28, 2008: National Cancer Institute ends Selenium and Vitamin E Cancer Prevention Trial, or SELECT
October 28, 2008: National Cancer Institute ends Selenium and Vitamin E Cancer Prevention Trial, or SELECT  March 16, 2008: New selenium-containing proteins identified in selenium-rich yeast
March 16, 2008: New selenium-containing proteins identified in selenium-rich yeast April 9, 2007: Trimethylselenonium is not the major metabolite
for human cancer patients excreting high doses of selenium
April 9, 2007: Trimethylselenonium is not the major metabolite
for human cancer patients excreting high doses of selenium  October 16, 2005: New light on human selenium metabolism
October 16, 2005: New light on human selenium metabolism October 6, 2005: Selenomethionine shows promising results as a protective agent against Esophageal Cancer
October 6, 2005: Selenomethionine shows promising results as a protective agent against Esophageal Cancer  August 2, 2005: New CRM for Selenomethionine in yeast developed by NRC Canada is now on the market
August 2, 2005: New CRM for Selenomethionine in yeast developed by NRC Canada is now on the market  March 8, 2005: Selenoprotein P is required for normal sperm development
March 8, 2005: Selenoprotein P is required for normal sperm developmentlast time modified: April 19, 2025