A US peer review panel has voted to accept the three key outcomes of a draft assessment of antimony trioxide that concludes the substance is 'reasonably anticipated' to be a human carcinogen.
Background:
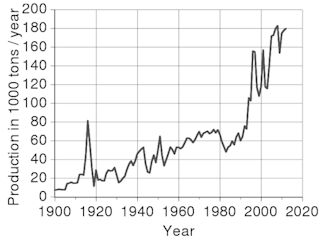
Figure: World production trend for antimony
(C) Leyo - U.S. Geological Survey |
Antimony substances are used extensively as flame retardants, and also
in lead batteries, plastics, paints, glass and ceramics, brake pads, semiconductors and ammunition.
Regulators have
been worried about their potential to cause lung cancer since the
1970s, when incidence among smelter workers was found to be higher than
for the general population. Antimony trioxide is typically used as a
proxy for the group, which includes antimony sulfide and antimony metal,
for the purposes of risk assessment.
The US National Toxicology Program (NTP) has recently finalized a two-year inhalation carcinogenicity study on Antimony Trioxide (ATO). Based on the draft results of this study, it appeared that the existing dataset on ATO would meet the three criteria which would justify a possible reclassification as carcinogen. The peer review panel consisting of six persons reviewed the draft
Report on Carcinogens (RoC) monograph from the National Toxicology
Program on 24 January.
Peer Review of NTP's draft RoC monograph:
According to the actions statement, they voted unanimously in favour of NTPs draft RoC monograph with respect to:
- the level of evidence conclusion that the data available from studies in humans are inadequate to evaluate the relationship between human cancer and exposure specifically to antimony trioxide or other antimony compounds;
- the level of evidence conclusion of sufficient evidence of carcinogenicity for antimony trioxide from studies in experimental animals; and
- the preliminary policy decision that antimony trioxide should be listed in the Report on Carcinogens as 'reasonably anticipated to be a human carcinogen' based on sufficient evidence from studies in experimental animals and supporting mechanistic data.
The animal data showed increases in the incidences of tumours in several organs of rats and mice exposed to antimony trioxide by inhalation. Enhanced number of tumors were observed for lung, skin and white blood cells in mice, and for lung and adrenal gland in rats. The review panel also agreed with the statement that a significant number of people living in the US are exposed to antimony trioxide.
The NTP received three comments during the consultation period. These came from a group of medical toxicology researchers at Drexel University College of Medicine in Philadelphia, Caroline Braibant at the International Antimony Association (i2A) and Campine, a Belgian company producing antimony products.
While the researchers from the Drexel University College of Medicine had only minor points of criticism, Ms Braibant from i2A said there were factual inaccuracies in the draft monograph and that she had significant reservations regarding:
- The summary statistics provided of current patterns of antimony trioxide consumption, use and occupational exposure.
- The absence of data defining particle size distributions in the occupational setting and comparative analysis of particle size effects in studies with experimental animals.
- Potentially erroneous extraction of data from the primary scientific literature.
- Conclusions that non-pulmonary neoplastic lesions in the NTP (2017) inhalation bioassays are specific effects induced by antimony trioxide.
- The validity of the genotoxicity studies conducted during the course of the recent NTP antimony trioxide inhalation bioassays.
Campine said that it routinely collected worker health data, under
Belgian legislation, which it had screened and subsequently drawn conclusions to them. The following conclusions have been made:
- Relationship between changes in pulmonary function parameters and years of exposure were weak.
- There were no clear relationships between mean urinary antimony concentration and
- changes in pulmonary function parameters
- liver function
- On the basis of the existing information of chest X-rays (on a yearly basis for more than 20 years) no pulmonary lesions were detected.
The company said it was preparing a publication based on the data and
asked the panel to delay its review to include the information.
The outcome of this review process could be significant in the EU, where the German REACH and
CLP competent authority, Bundesanstalt für Arbeitsschutz und Arbeitsmedizin (BAuA), is to begin evaluating antimony trioxide
along with antimony sulfide and antimony metal in March. Industry is
concerned that the results could lead to stricter regulation.
 Related information
Related information
 NIEHS/NIH: National Toxicology Program: Monograph on Antimony Trioxide, October 2018
NIEHS/NIH: National Toxicology Program: Monograph on Antimony Trioxide, October 2018  International Antimony Association: February 21, 2020: i2a observations on the October 2019 ATSDR Toxicological Profile for Antimony and Antimony Compounds
International Antimony Association: February 21, 2020: i2a observations on the October 2019 ATSDR Toxicological Profile for Antimony and Antimony Compounds
 INCHEM: Toxicity of Antimony Trioxide
INCHEM: Toxicity of Antimony Trioxide  IARC: Monography on Antimony Trioxide and Antimony Trisulfide
IARC: Monography on Antimony Trioxide and Antimony Trisulfide  NIOSH: Toxicity of Antimony Compounds
NIOSH: Toxicity of Antimony Compounds
 Related EVISA Resources
Related EVISA Resources
 Link database: Toxicity of antimony
Link database: Toxicity of antimony
 Link database: Industrial use of antimony
Link database: Industrial use of antimony
 Related News
Related News April 14, 2010: Antimony mine disaster
April 14, 2010: Antimony mine disaster  September 18,2008: REACH Update: List of 300 chemicals of very high concern
September 18,2008: REACH Update: List of 300 chemicals of very high concern
 August 16, 2006: Toxic antimony species found in beverages stored in PET containers
August 16, 2006: Toxic antimony species found in beverages stored in PET containers
last time modified: November 27, 2023