Governmental health departments all over the world have decided to recommend the injection of an organic mercury compound directly into the bodies of their inhabitants. This field trial will start in a few days, will have millions of participants all over the globe and is especially targeting at pregnant women, infants and the vulnerable elder people.
Background:Mercury is a neurotoxic heavy metal that is especially dangerous for developing infants. In the form of organic mercury compounds it even can cross the placenta and the blood/brain barrier and therefore is considered one of the strongest neurotoxin for humans and animals. Due to this fact, governments all over the globe but especially in Europe and the USA have started activities to reduce the risk for their population and especially vulnerable groups such as pregnant women and infants. These activities are targeting at the reduction of mercury emission to the environment, by phasing out the use of mercury in consumer goods where it can easily be substituted (thermometers, batteries, electrical switches), by reducing the industrial use of mercury (e.g. production of chlorine) and by reducing emissions from power plants.
Especially vulnerable groups (pregnant women) are advised how to reduce their mercury uptake by controlling their diet with respect to seafood containing mercury (see the
mercury warning of EPA/FDA in the USA).
The new study:
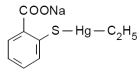
While the toxicity of mercury and especially methyl-mercury is well known (see the resources below), the data available for ethylmercury is more sparingly. That is the reason that many govern-mental health departments have agreed to authorize a field experiment which foresees the injection of an ethylmercury containing compound called thimerosal or thiomersal directly into the bodies of their inhabitants.
Figure: Formula of Thimerosal (thiomersal)
You do not believe such news? Indeed, you might wonder: If a research group of toxicologists would have suggested this field experiment, no ethic committee would have authorized it - that is for sure. However this field trial was organized by the health authorities themselves and is called
swine flue vaccination campaign.
The facts:
Regulators in Europe and the United States plan to fast-track approval of swine flu vaccines to ensure they are available for the start of the northern hemisphere winter. In the middle of next months the vaccine against the swine flue will be distributed by the manufacturers. Leading flu vaccine makers including CSL Ltd. of Australia, Switzerland's Novartis Vaccines, Maryland-based MedImmune LLC, Sanofi Pasteur of France, London-based GlaxoSmithKline, Baxter and China-based Sinovac rush to share the expected multi-billion dollar business by bringing their products to the market before the winter season starts.
WHO currently estimates worldwide production capacity for pandemic vaccines at approximately 3 billion doses per year (see WHO: H1N1 briefing note 11).Company
| Product
| Doses scheduled
for 2009
| Adjuvants/Preservation
(see the product info)
| Countries
served
|
CSL
| Panvax
| ??
| thimerosal
| USA, Australia
|
Novartis
| Celtura, Focetria
| 150 Mio
| squalene, thimerosal
| USA |
MedImmune (AstraZeneca)
|
| 12 Mio
| none
| USA
|
Sanofi
| Fluzone
| 800 Mio
| thimerosal
| USA, France
|
GlaxoSmithKline
| Pandemrix
| 50 Mio (DE)
| squalene, thimerosal
| Germany, Canada
|
Baxter
| Celvapan
| 80 Mio
| none
| UK, Ireland, New Zealand
|
Sinovac
|
| 3.3 Mio
| ??
| China
|
There are a few controversial ingredients added to the raw vaccine called adjuvants, meant to boost the immune reaction. Some of the products will contain squalene as adjuvant. Squalene, a biochemical/cholesterol precursor to the family of steroids, stimulates the immune system nonspecifically. While these adjuvants are commonly used in European flu vaccines, there are no licensed flu vaccines in the U.S. or Canada with the ingredients and there is limited data on how safe flu vaccines with adjuvants are in pregnant women and children
. Today, as we are marketing to these vary populations, WHO says
As this vaccine and new squalene-containing vaccines are introduced in other age groups, post-marketing follow-up to detect any vaccine-related adverse events will need to be performed.
Such kind of approach is simply "a large-scale experiment being
conducted on the ... population," warns Wolfgang Becker-Brüser, the
publisher of
arznei telegramm, a professional journal critical of the pharmaceutical industry.
Another compound added to the raw vaccine is Thimerosal, an ethylmercury containing compound, used as a preservative. Since this component was introduced in 1931, it has never gone through any meaningful evaluation and due to its toxicity profile probably would not be approved for any testing today. Because of the risk associated with the neurotoxin, the use of thimerosal was phased out in Europe and the USA since 1999 and has been banned for over-the-counter medicines. Unfortunately as an adjuvant in vaccines it was only phased out by industry because of pressure from the public but never banned by regulation. The FDA in 1999 made a recommendation to manufacturers to remove the mercury from vaccines on the CDC's recommended schedule for ages birth to three when they realized that children were receiving a cumulative dose of mercury that was potentially harmful. They did not recall the mercury-containing vaccines, nor were they banned. Instead, they allowed existing stocks to be used up, which took several years. Note that the FDA's directive never applied to vaccines not on the CDC's recommended childhood schedule (for example, Diphtheria/Tetanus). Some states (e.g. Iowa, California, Delaware, Illinois, New York, Missouri and Washington)
currently have enacted state laws that protect vulnerable populations from
receiving thimerosal-containing vaccines but enforcement is inconsistent. Now it is coming back in many swine flu vaccines (see the table) in typical concentrations of 50 µg/ml (50 ppm).
CSL's Dr Rachel David, said thiomersal was not banned in Australia and was actually
beneficial. "The reason it is in the multi-dose vials is to prevent
contamination, so there is a very real benefit from using it," Dr David
said. US EPA says that safety limits are 0.1 µg per kg of body weight per day, which makes this vaccine (a single shot is 0.5 ml) safe for anyone who weights 250 kg !
In order to ensure that sufficient doses of the vaccine are available,
some states (e.g. Washington) have temporarily suspended their state laws limiting the amount of mercury (thimerosal) allowed in H1N1 (swine flu)
vaccine given to pregnant women and children under three.
In case that the field testing will show problems, the poor victims will probably find it problematic to receive compensation. In the US vaccine makers and federal officials will be immune from lawsuits that result from any new swine flu vaccine, under a document signed by Secretary of Health and Human Services Kathleen Sebelius (
see the news below). The document signed by Sebelius last month grants immunity to those making a swine flu vaccine, under the provisions of a 2006 law for public health emergencies. Other governments also have signed secret contracts. Australian Health Minister Nicola Roxon said "There are some confidentiality issues. We have
a private contractual agreement with CSL for the priority provision for
this vaccine".
At least the risk due to thimerosal is avoidable, since it may be possible to find vaccine that only contain small amounts of thimerosal (about 1 µg thimerosal per dose) called thimerosal-free, if you ask for single-dose shots. According to the CDC, "Single-dose syringes will be thimerosal-free, which will address concerns about this additive, especially regarding pediatric and pregnant vaccine recipients (inhaler sprayer vaccine products will also be thimerosal-free)."
As it is written in the package info of the vaccines, "the decision to give Influenza A (H1N1) ...vaccine should be based on careful consideration of the potential benefits and risks".
Michael Sperling
 Studies related to health risks imposed by thimerosal
Studies related to health risks imposed by thimerosal
D.A. Geier, P.G. King, M.R. Geier,
Mitochondrial dysfunction, impaired oxidative-reduction activity, degeneration, and death in human neuronal and fetal cells induced by low-level exposure to thimerosal and other metal compounds, Toxicol. Environ. Chem., 91/4 (2009) 735-749.
DOI: 10.1080/02772240802246458
Alberto Eugenio Tozzi, Patrizia Bisiacchi, Vincenza Tarantino, Barbara De Mei, Lidia D'Elia, Flavia Chiarotti, Stefania Salmaso,
Neuropsychological Performance 10 Years After Immunization in Infancy with Thimerosal-Containing Vaccines, Pediatrics, 123/2 (2009) 475-482.
DOI: 10.1542/peds.2008-0795

Donald R. Branch,
Gender selective toxicity of thimerosal, Exp. Toxicol. Pathol., 61/2 (2009) 133-136.
DOI: 10.1016/j.etp.2008.07.002
José G. Dórea, Rejane C. Marques,
Modeling Neurodevelopment Outcomes and Ethylmercury Exposure from Thimerosal-Containing Vaccines, Toxicol. Sci., 103/2 (2008) 414-415.
DOI: 10.1093/toxsci/kfn049
Grazyna Zareba, Elsa Cernichiari, Rieko Hojo, Scott Mc Nitt, Bernard Weiss, Moiz M. Mumtaz, Dennis E. Jones, Thomas W. Clarkson,
Thimerosal distribution and metabolism in neonatal mice: comparison with methyl mercury, J. Appl. Toxicol., 27/5 (2007) 511-518.
DOI: 10.1002/jat.1272
David A. Geier, Mark R. Geier,
Early Downward Trends in Neurodevelopmental Disorders Following Removal of Thimerosal-Containing Vaccines, J. Am. Phys. Surg., 11/1 (2006) 8-13.

David A. Geier, Mark R. Geier,
A two-phased population epidemiological study of the safety of thimerosal-containing vaccines: a follow-up analysis,
Med. Sci. Monit., 11/4 (2005) CR160-170. 
Mark F. Blaxill, Lyn Redwood, Sallie Bernard,
Thimerosal and autism? A plausible hypothesis that should not be dismissed, Med. Hypotheses, 62 (2004) 788-794.
DOI: 10.1016/j.mehy.2003.11.033
Thomas Verstraeten, Robert L. Davis, Frank DeStefano, Tracy A. Lieu, Philip H. Rhodes, Steven B. Black, Henry Shinefield, Robert T. Chen,
Safety of Thimerosal-Containing Vaccines: A Two-Phased Study of Computerized Health Maintenance Organization Databases, Pediatrics, 112/5 (2003) 1039-1048. available at:
http://pediatrics.aappublications.org/cgi/reprint/112/5/1039 Related information
Related information Wikipedia: Swine influenza
Wikipedia: Swine influenza Wikipedia: 2009 flu pandemic
Wikipedia: 2009 flu pandemic Wikipedia: Thimerosal
Wikipedia: Thimerosal CDC: Thimerosal in Seasonal Influenza Vaccine
CDC: Thimerosal in Seasonal Influenza Vaccine CDC: 2009 H1N1 Flu (Swine Flu) and You
CDC: 2009 H1N1 Flu (Swine Flu) and You WHO: Gloabal Alert and Response: Pandemic (H1N1) 2009
WHO: Gloabal Alert and Response: Pandemic (H1N1) 2009 WHO: (H1N1) 2009 briefing note 7: Pandemic influenza vaccine manufacturing process and timeline
WHO: (H1N1) 2009 briefing note 7: Pandemic influenza vaccine manufacturing process and timeline Paul-Ehrlich-Insitut (in German): Comments of Experts on the Risk from H1N1
Paul-Ehrlich-Insitut (in German): Comments of Experts on the Risk from H1N1 European Commission: Public Health: Pandemic (H1N1) 2009
European Commission: Public Health: Pandemic (H1N1) 2009
 Related EVISA Resources
Related EVISA Resources Link Database: Toxicity of organic mercury
Link Database: Toxicity of organic mercury EVISA News about Thimerosal
EVISA News about Thimerosal EVISA Advanced Search: All about Thimerosal
EVISA Advanced Search: All about Thimerosal
 Related EVISA News
Related EVISA News
last time modified: June 29, 2020