Selenomethionine treatment of certain high-risk patients show promise as protection against the development of esophageal squamous cell cancer (ESCC), according to a report in the September issue of Gastroenterology.
Esophageal squamous cell carcinoma remains a leading cause of cancer death worldwide. Squamous dysplasia, the accepted histological precursor for esophageal squamous cell carcinoma, represents a potentially modifiable intermediate end point for chemoprevention trials in high-risk populations.
While selenium compounds and COX-2 inhibitors have shown potential chemopreventive effects on esophageal carcinogenesis in previous observational studies, such effects had not been evaluated directly in ESCC chemoprevention trials, as pointed out by the authors of the study. Dr. Paul J. Limburg from Mayo Clinic College of Medicine, Rochester, Minnesota and his colleagues now filled this gap by testing the chemoprevention potential of selenomethionine (a synthetic form of organic selenium) and celecoxib (a selective inhibitor of COX-2) in 238 asymptomatic adults with histologically confirmed mild or moderate esophageal squamous dysplasia.
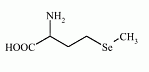
Figure: selenomethionine
|
In the overall analysis of the 10-month intervention, neither selenomethionine nor celecoxib significantly changed the dysplasia grade during the course of the trial. However, patients treated with selenomethionine showed a trend toward increased regression and decreased progression, the report indicates, but no such trend was seen with celecoxib treatment.
In a secondary, stratified analysis, treatment of 115 patients with mild dysplasia with selenomethionine was associated with nearly a doubling of the regression rate and halving of the dysplasia progression rate, compared with placebo. No apparent chemopreventive effect was seen in the 123 patients with moderate dysplasia.
The authors conclude that use of celecoxib appears to lack promise but selenomethionine did have a protective effect among subjects with mild esophageal squamous dysplasia at baseline.
 The original study:
The original study:

Paul J. Limburg, Wenqiang Wei, Dennis J. Ahnen, Youlin Qiao, Ernest T. Hawk, Guoqing Wang, Carol A. Giffen, Guiqi Wang, Mark J. Roth, Ning Lu, Edward L. Korn, Yurong Ma, Kathleen L. Caldwell, Zheiwei Dong, Philip R. Taylor, Sanford M. Dawsey,
Randomized, Placebo-Controlled, Esophageal Squamous Cell Cancer Chemoprevention Trial of Selenomethionine and Celecoxib, Gastroenterology, 129 (2005) 863-873. DOI:
10.1053/j.gastro.2005.06.024
 Related Studies
Related Studies

F. Fernandez-Banares, E. Cabre, M. Esteve, M.D. Mingorance, A.
Abad-La M. Lachica, A. Gil, M.A. Gassull,
Serum selenium and risk of
large size colorectal adenomas in a geographic area with a low selenium
status, Am. J. Gastroenterol., 97/8 (2002) 2103-2108.
DOI:
10.1111/j.1572-0241.2002.05930.x

H.E. Ganther,
Selenium metabolism and mechanism of cancer prevention, Adv. Exp. Med. Biol., 492 (2001) 119-130.
DOI:
10.1007/978-1-4615-1283-7_10

C. Ip,
Lessons from Basic Research in Selenium and Cancer Prevention, J. Nutr. , 128 (1998) 1845-1854.
DOI:
10.1093/jn/128.11.1845

G.F. Combs, L.C. Clark, B.W. Turnbull,
Reduction of cancer risk with
oral supplement of selenium, Biomed. Environ. Sci., 10 (1997) 227-234.
PMID:
9315315

L.C. Clark, G.F. Combs, B.W. Turnbull, E.H. Slate, D.K. Chalker, J.
Chow, L.S. Davis, R.E. Glover, G.F. Graham, E.G. Gross, A. Krongard, et
al.,
Effects of Selenium Supplementation for Cancer Prevention in
Patients With Carcinoma of the Skin, J. Am. Med., 276/24 (1996)
1957-1963.
DOI: 10.1001/jama.1996.03540240035027

K. Overvad, D.Y. Wang, J. Olsen, D.S. Allen, E.B. Thorling, R.D. Bulbrook, J.L. Hayward,
Selenium in human mammary carcinogenesis: a case cohort study, Eur. J. Cancer, 27 (1991) 900-902.
DOI: 10.1016/0277-5379(91)90143-2

J.T. Salonen, G. Alfthan, J.K. Huttunen, P. Puska,
Association between serum selenium and the risk of cancer, Am. J. Epidemiol., 120 (1984) 342-349.
DOI:
10.1093/oxfordjournals.aje.a113898

A.M. Watrach, J.A. Milner, M.A. Watrach, K.A. Poirier,
Inhibition of human breast cancer cells by selenium, Cancer Letter (Shannon, Irel.), 25 (1984) 41-47. DOI:
10.1016/S0304-3835(84)80024-5
 Related EVISA Resources
Related EVISA Resources
 Link Database: Selenium and Human health
Link Database: Selenium and Human health
 Link Database; Research projects related to selenium
Link Database; Research projects related to selenium
 Link database: All about proteins containing selenium
Link database: All about proteins containing selenium
 Related EVISA News (newest first)
Related EVISA News (newest first)
 August 16, 2016: Toxicity and bioavailability of different selenium metabolites
August 16, 2016: Toxicity and bioavailability of different selenium metabolites
 July 21, 2015: Polish selenium supplements not always labeled accurately
July 21, 2015: Polish selenium supplements not always labeled accurately
 December 17, 2014: Decreased risk of colorectal cancer linked with higher selenium status
December 17, 2014: Decreased risk of colorectal cancer linked with higher selenium status
 August 9, 2011: New selenium metabolites found in human serum
August 9, 2011: New selenium metabolites found in human serum May 22, 2011: Does Selenium Prevent Cancer? It May Depend on Which Form People Take
May 22, 2011: Does Selenium Prevent Cancer? It May Depend on Which Form People Take
 May 12, 2011: Review: Selenium doesn't prevent cancer
May 12, 2011: Review: Selenium doesn't prevent cancer June 19, 2010: A
new Selenium-containing compound, Selenoneine, found as the predominant
Se-species in the blood of Bluefin Tuna
June 19, 2010: A
new Selenium-containing compound, Selenoneine, found as the predominant
Se-species in the blood of Bluefin Tuna  July 20, 2009: Researchers Reveal Selenium's Metabolism In Life-Giving Amino Acids
July 20, 2009: Researchers Reveal Selenium's Metabolism In Life-Giving Amino Acids October 28, 2008: National Cancer Institute ends Selenium and Vitamin E Cancer
Prevention Trial, or SELECT
October 28, 2008: National Cancer Institute ends Selenium and Vitamin E Cancer
Prevention Trial, or SELECT March 16, 2008: New selenium-containing proteins identified in selenium-rich yeast
March 16, 2008: New selenium-containing proteins identified in selenium-rich yeast October 16, 2005: New light on human selenium metabolism
October 16, 2005: New light on human selenium metabolism October 6, 2005: Selenomethionine shows promising results as a protective agent
against Esophageal Cancer
October 6, 2005: Selenomethionine shows promising results as a protective agent
against Esophageal Cancer
 August 2, 2005: New CRM for Selenomethionine in yeast developed by NRC Canada is now on the market
August 2, 2005: New CRM for Selenomethionine in yeast developed by NRC Canada is now on the market
last time modified: March 6, 2024