By replacing sulfur with selenium, scientists have developed a long-lasting synthetic insulin analogue that could be used for treating diabetes.
Background:Diabetes mellitus (DM) is caused by either the pancreas not producing enough insulin (Type 1 DM) or the cells of the body not responding properly to the insulin produced (Type 2 DM) resulting in the bodys inability to control blood sugar levels. As a result, diabetics have to consistently monitor their blood glucose concentration, alter their diet and lifestyles, and may have to administer insulin injections to regulate the amount of free sugar in their blood. As of 2015, an estimated 415 million people had diabetes worldwide, with type 2 DM making up about 90% of the cases.
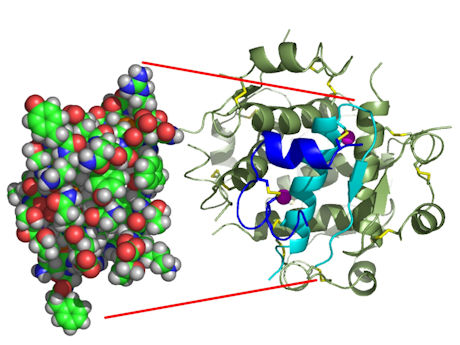
Figure: The structure of insulin. The left side is a space-filling model of the insulin monomer, believed to be biologically active. Carbon is green, hydrogen white, oxygen red, and nitrogen blue. On the right side is a ribbon diagram of the insulin hexamer, believed to be the stored form. A monomer unit is highlighted with the A chain in blue and the B chain in cyan. Yellow denotes disulfide bonds, and magenta spheres are zinc ions.
A group of researchers in Japan has succeeded in synthesizing an insulin analogue with long-lasting activity by replacing the interchain disulfide in bovine pancreatic insulin (BPIns) with a diselenide bridge. The researchers suggested that the introduction of two juxtaposed selenium atoms to the insulin analogue could lead to a higher kinetic and thermodynamic stability than that of the wild-type without affecting the bioactivity. Indead, the synthesized selenoinsulin exhibited a bioactivity comparable to that of BPIns and its degradation was significantly decelerated (τ1/2≈8 h vs. ≈1 h for BPIns).
 The original study:
The original study:

Kenta Arai, Toshiki Takei, Masaki Okumura, Satoshi Watanabe, Yuta Amagai, Yuya Asahina, Luis Moroder, Hironobu Hojo, Kenji Inaba, Michio Iwaoka,
Preparation of Selenoinsulin as a Long-Lasting Insulin Analogue, Angew. Chem. Int. Ed. 56 (2017) 16.
DOI: 10.1002/anie.201701654. Related studies
Related studies

A.P. Ryle, F. Sanger, L.F. Smith, Ruth Kitai,
The disulphide bonds of insulin, Biochem. J. 60/4 (1955) 541-556;
DOI: 10.1042/bj0600541
Joseph M.Tibaldi,
Evolution of Insulin: From Human to Analog, Am. J. Med., 127/10 Supplement (2014) S25-S38.
DOI: 10.1016/j.amjmed.2014.07.005
W.C. Duckworth, R.G. Bennett, F.G. Hamel,
Insulin degradation: progress and potential, Endocr. Rev. 19/5 (1998) 608-624.
DOI: 10.1210/edrv.19.5.0349
W.J. Tang,
Targeting Insulin-Degrading Enzyme to Treat Type 2 Diabetes Mellitus, Trends Endocrinol. Metabol., 27/1 (2016) 24-34.
DOI: 10.1016/j.tem.2015.11.003
Safia Costes, Peter C. Butler,
Insulin-Degrading Enzyme Inhibition, a Novel Therapy for Type 2 Diabetes?, Cell Metabolism, 20/2 (2014) 201-203.
DOI: 10.1016/j.cmet.2014.07.016
J.A. Karas, N.A. Patil, J. Tailhades, M.A. Sani, D.B. Scanlon, B.E. Forbes, J. Gardiner, F. Separovic, J.D. Wade, M.A. Hossain,
Total Chemical Synthesis of an Intra-A-Chain Cystathionine Human Insulin Analogue with Enhanced Thermal Stability, Angew. Chem. Int. Ed. Engl., 55/47 (2016) 14743-14747.
DOI: 10.1002/anie.201607101
Luis Moroder,
Isosteric replacement of sulfur with other chalcogens in peptides and proteins, J. Pep. Sci., 11/4 (2006) 187-214.
DOI: 10.1002/psc.654
Marika Manolopoulou, Qing Guo, Enrico Malito, Alexander B. Schilling, Wei-Jen Tang,
Molecular Basis of Catalytic Chamber-assisted Unfolding and Cleavage of Human Insulin by Human Insulin-degrading Enzyme, J. Biol. Chem., 284 (2009) 1417714188.
 Related EVISA News
Related EVISA News
 October 15, 2016: Researchers discover how selenium is incorporated into proteins
October 15, 2016: Researchers discover how selenium is incorporated into proteins
last time modified: August 20, 2017